- Anthracycline
- A member of a family of chemotherapy drugs that are also antibiotics. The anthracyclines act to prevent cell division by disrupting the structure of the DNA and terminate its function. They do so in two ways: (1) they intercalate into the base pairs in the DNA minor grooves; and (2) they cause free radical damage of the ribose in the DNA. The anthracyclines are frequently used in leukemia therapy. The anthracyclines include daunorubicin (Cerubidine), doxorubicin (Adriamycin, Rubex), epirubicin (Ellence, Pharmorubicin), and idarubicin (Idamycin).
* * *Anticancer agent consisting of 3 moieties: a pigmented aglycone, an amino sugar, and a lateral chain. Examples are doxorubicin, daunorubicin, and daunomycin.
* * *
an·thra·cy·cline .an(t)-thrə-'sī-.klēn n any of a class of antineoplastic drugs (as doxorubicin) derived from an actinomycete of the genus Streptomyces (esp. S. peucetius)* * *
n.any of 500 or so antibiotics synthesized or isolated from species of Streptomyces. doxorubicin is the most important member of this group of compounds, which have wide activity against tumours.* * *
an·thra·cy·cline (an″thrə-siґklēn) any of a class of antineoplastic antibiotics, including daunorubicin and doxorubicin, produced by Streptomyces peucetius or S. coeruleorubidus and having a four-ring system to which a daunosamine molecule is attached by glycoside linkage (see illustration). Although the precise mechanism of action is unknown, anthracyclines can damage DNA by intercalation, metal ion chelation, and the generation of free radicals and can inhibit enzyme activity critical to DNA function. The use of these drugs is limited by dose-related cardiotoxicity.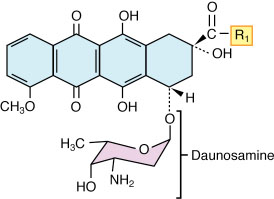
 Anthracycline. For daunorubicin, R1 =
Anthracycline. For daunorubicin, R1 =  CH3; for doxorubicin, R1 =
CH3; for doxorubicin, R1 =  CHOH.
CHOH.
Medical dictionary. 2011.
